Ubiquitin ligase
| Ubiquitin—protein ligase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 E3 ubiquitin ligase Cbl (blue) in complex with E2 (cyan) and substrate peptide (green). PDB entry 4a4c[1] | |||||||||
| Identifiers | |||||||||
| EC number | 2.3.2.27 | ||||||||
| CAS number | 74812-49-0 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Ubiquitin ligase | |
|---|---|
| Identifiers | |
| Symbol | Ubiquitin ligase |
| OPM family | F471 |
| OPM protein | 4v6p |
| Membranome | 240 |
A ubiquitin ligase (also called an E3 ubiquitin ligase) is a protein that recruits an E2 ubiquitin-conjugating enzyme that has been loaded with ubiquitin, recognizes a protein substrate, and assists or directly catalyzes the transfer of ubiquitin from the E2 to the protein substrate. The ubiquitin is attached to a lysine on the target protein by an isopeptide bond.[2] E3 ligases interact with both the target protein and the E2 enzyme, and so impart substrate specificity to the E2. Commonly, E3s polyubiquitinate their substrate with Lys48-linked chains of ubiquitin, targeting the substrate for destruction by the proteasome. However, many other types of linkages are possible and alter a protein's activity, interactions, or localization. Ubiquitination by E3 ligases regulates diverse areas such as cell trafficking, DNA repair, and signaling and is of profound importance in cell biology. E3 ligases are also key players in cell cycle control, mediating the degradation of cyclins, as well as cyclin dependent kinase inhibitor proteins.[3] The human genome encodes over 600 putative E3 ligases, allowing for tremendous diversity in substrates.[4]
Contents
1 Overview
2 Ubiquitination system
3 Ubiquitin ligase families
4 Mono- and poly- ubiquitylation
5 Disease relevance
5.1 Examples
6 Individual E3 ubiquitin ligases
7 See also
8 References
9 External links
Overview
In enzymology, an ubiquitin-protein ligase (EC 6.3.2.19) is an enzyme that catalyzes the chemical reaction
- ATP + ubiquitin + protein lysine ⇌{displaystyle rightleftharpoons }
AMP + diphosphate + protein N-ubiquityllysine
The 3 substrates of this enzyme are ATP, ubiquitin, and a lysine residue on a protein, whereas its 3 products are AMP, diphosphate, and protein N-ubiquityllysine. Canonical ubiquitylation creates an isopeptide bond between a lysine residue on a target protein and the ubiquitin C-terminal Glycine 76.[5]
This enzyme belongs to the family of ligases, to be specific those forming carbon-nitrogen bonds as acid-D-amino-acid ligases (peptide synthases). The systematic name of this enzyme class is ubiquitin:protein-lysine N-ligase (AMP-forming). This enzyme is also called ubiquitin-activating enzyme. This enzyme participates in 3 metabolic pathways: ubiquitin-mediated proteolysis, Parkinson's disease, and Huntington's disease.[citation needed]
Ubiquitination system
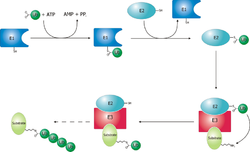
Schematic diagram of the ubiquitylation system.
The ubiquitin ligase is referred to as an E3, and operates in conjunction with an E1 ubiquitin-activating enzyme and an E2 ubiquitin-conjugating enzyme. There is one major E1 enzyme, shared by all ubiquitin ligases, that uses ATP to activate ubiquitin for conjugation and transfers it to an E2 enzyme. The E2 enzyme interacts with a specific E3 partner and transfers the ubiquitin to the target protein. The E3, which may be a multi-protein complex, is, in general, responsible for targeting ubiquitination to specific substrate proteins.[citation needed]
The ubiquitylation reaction proceeds in three or four steps depending on the mechanism of action of the E3 ubiquitin ligase. In the conserved first step, an E1 cysteine residue attacks the ATP-activated C-terminal glycine on ubiquitin, resulting in a thioester Ub-S-E1 complex. The energy from ATP and diphosphate hydrolysis drives the formation of this reactive thioester, and subsequent steps are thermoneutral. Next, a transthiolation reaction occurs, in which an E2 cysteine residue attacks and replaces the E1. HECT domain type E3 ligases will have one more transthiolation reaction to transfer the ubiquitin molecule onto the E3, whereas the much more common RING finger domain type ligases transfer ubiquitin directly from E2 to the substrate.[6] The final step in the first ubiquitylation event is an attack from the target protein lysine amine group, which will remove the cysteine, and form a stable isopeptide bond.[5] One notable exception to this is p21 protein, which appears to be ubiquitylated using its N-terminal amine, thus forming a peptide bond with ubiquitin.[7]
Ubiquitin ligase families
Humans have an estimated 500-1000 E3 ligases, which impart substrate specificity onto the E1 and E2.[8] The E3 ligases are classified into four families: HECT, RING-finger, U-box, and PHD-finger.[8] The RING-finger E3 ligases are the largest family and contain ligases such as the anaphase-promoting complex (APC) and the SCF complex (Skp1-Cullin-F-box protein complex). SCF complexes consist of four proteins: Rbx1, Cul1, Skp1, which are invariant among SCF complexes, and an F-box protein, which varies. Around 70 human F-box proteins have been identified.[9] F-box proteins contain an F-box, which binds the rest of the SCF complex, and a substrate binding domain, which gives the E3 its substrate specificity.[8]
Mono- and poly- ubiquitylation

Ubiquitin with lysine residues (red), N-terminal methionine (blue), and C-terminal glycine (yellow).[10]
Ubiquitin signaling relies on the diversity of ubiquitin tags for the specificity of its message. A protein can be tagged with a single ubiquitin molecule (monoubiquitylation), or variety of different chains of ubiquitin molecules (polyubiquitylation).[11] E3 ubiquitin ligases catalyze polyubiquitination events much in the same way as the single ubiquitylation mechanism, using instead a lysine residue from a ubiquitin molecule currently attached to substrate protein to attack the C-terminus of a new ubiquitin molecule.[5][11] For example, a common 4-ubiquitin tag, linked through the lysine at position 48 (K48) recruits the tagged protein to the proteasome, and subsequent degradation.[11] However, all seven of the ubiquitin lysine residues (K6, K11, K27, K29, K48, and K63), as well as the N-terminal methionine are used in chains in vivo.[11]
Monoubiquitination has been linked to membrane protein endocytosis pathways. For example, phosphorylation of the Tyrosine at position 1045 in the Epidermal Growth Factor Receptor (EGFR) can recruit the RING type E3 ligase c-Cbl, via an SH2 domain. C-Cbl monoubiquitylates EGFR, signaling for its internalization and trafficking to the lysosome.[12]
Monoubiquitination also can regulate cytosolic protein localization. For example, the E3 ligase MDM2 ubiquitylates p53 either for degradation (K48 polyubiquitin chain), or for nuclear export (monoubiquitylation). These events occur in a concentration dependent fashion, suggesting that modulating E3 ligase concentration is a cellular regulatory strategy for controlling protein homeostasis and localization.[13]
Disease relevance
E3 ubiquitin ligases regulate homeostasis, cell cycle, and DNA repair pathways, and as a result, a number of these proteins are involved in a variety of cancers, including famously MDM2, BRCA1, and Von Hippel-Lindau tumor suppressor.[14] For example, a mutation of MDM2 has been found in stomach cancer,[15]renal cell carcinoma,[16] and liver cancer [17] (amongst others) to deregulate MDM2 concentrations by increasing its promoter’s affinity for the Sp1 transcription factor, causing increased transcription of MDM2 mRNA.[15]
Examples
- A RING (Really Interesting New Gene) domain binds the E2 conjugase and might be found to mediate enzymatic activity in the E2-E3 complex[18]
- An F-box domain (as in the SCF complex) binds the ubiquitinated substrate. (e.g., Cdc 4, which binds the target protein Sic1; Grr1, which binds Cln).[19]
- A HECT domain, which is involved in the transfer of ubiquitin from the E2 to the substrate.
Individual E3 ubiquitin ligases
- E3A
- mdm2
Anaphase-promoting complex (APC)
UBR5 (EDD1)
SOCS/ BC-box/ eloBC/ CUL5/ RING
- LNXp80
CBX4, CBLL1
- HACE1
HECTD1, HECTD2, HECTD3, HECTD4
HECW1, HECW2
HERC1, HERC2, HERC3, HERC4, HERC5, HERC6
HUWE1, ITCH
NEDD4, NEDD4L
- PPIL2
- PRPF19
PIAS1, PIAS2, PIAS3, PIAS4
- RANBP2
- RNF4
- RBX1
SMURF1, SMURF2
- STUB1
- TOPORS
- TRIP12
UBE3A, UBE3B, UBE3C, UBE3D
UBE4A, UBE4B
- UBOX5
- UBR5
WWP1, WWP2
- Parkin
See also
- ERAD
- Ubiquitin
- Ubiquitin-activating enzyme
- Ubiquitin-conjugating enzyme
References
^ Dou H, Buetow L, Hock A, Sibbet GJ, Vousden KH, Huang DT (Feb 2012). "Structural basis for autoinhibition and phosphorylation-dependent activation of c-Cbl". Nature Structural & Molecular Biology. 19 (2): 184–92. doi:10.1038/nsmb.2231. PMC 3880865. PMID 22266821..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output .citation q{quotes:"""""""'""'"}.mw-parser-output .citation .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .citation .cs1-lock-limited a,.mw-parser-output .citation .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .citation .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-ws-icon a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/4/4c/Wikisource-logo.svg/12px-Wikisource-logo.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-maint{display:none;color:#33aa33;margin-left:0.3em}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ Hershko A, Ciechanover A (1998). "The ubiquitin system". Annual Review of Biochemistry. 67: 425–79. doi:10.1146/annurev.biochem.67.1.425. PMID 9759494.
^ Teixeira LK, Reed SI (2013). "Ubiquitin ligases and cell cycle control". Annual Review of Biochemistry. 82: 387–414. doi:10.1146/annurev-biochem-060410-105307. PMID 23495935.
^ Li W, Bengtson MH, Ulbrich A, Matsuda A, Reddy VA, Orth A, Chanda SK, Batalov S, Joazeiro CA (2008). "Genome-wide and functional annotation of human E3 ubiquitin ligases identifies MULAN, a mitochondrial E3 that regulates the organelle's dynamics and signaling". PLoS ONE. 3 (1): e1487. Bibcode:2008PLoSO...3.1487L. doi:10.1371/journal.pone.0001487. PMC 2198940. PMID 18213395.
^ abc Walsh, Christopher (2006). Posttranslational Modification of Proteins: Expanding Nature's Inventory. Englewood, CO: Roberts. ISBN 978-0-9747077-3-0.
[page needed]
^ Metzger MB, Hristova VA, Weissman AM (Feb 2012). "HECT and RING finger families of E3 ubiquitin ligases at a glance". Journal of Cell Science. 125 (Pt 3): 531–7. doi:10.1242/jcs.091777. PMC 3381717. PMID 22389392.
^ Bloom J, Amador V, Bartolini F, DeMartino G, Pagano M (Oct 2003). "Proteasome-mediated degradation of p21 via N-terminal ubiquitinylation". Cell. 115 (1): 71–82. doi:10.1016/S0092-8674(03)00755-4. PMID 14532004.
^ abc Nakayama KI, Nakayama K (May 2006). "Ubiquitin ligases: cell-cycle control and cancer". Nature Reviews. Cancer. 6 (5): 369–81. doi:10.1038/nrc1881. PMID 16633365.
^ Jin J, Cardozo T, Lovering RC, Elledge SJ, Pagano M, Harper JW (Nov 2004). "Systematic analysis and nomenclature of mammalian F-box proteins". Genes & Development. 18 (21): 2573–80. doi:10.1101/gad.1255304. PMC 525538. PMID 15520277.
^ Vijay-Kumar, S.; Bugg, C. E.; Cook, W. J. (1987). "Structure of ubiquitin refined at 1.8 A resolution". J. Mol. Biol. 194 (3): 531–44. doi:10.1016/0022-2836(87)90679-6. PMID 3041007.
^ abcd Behrends C, Harper JW (May 2011). "Constructing and decoding unconventional ubiquitin chains". Nature Structural & Molecular Biology. 18 (5): 520–8. doi:10.1038/nsmb.2066. PMID 21540891.
^ Bonifacino JS, Traub LM (2003). "Signals for sorting of transmembrane proteins to endosomes and lysosomes". Annual Review of Biochemistry. 72: 395–447. doi:10.1146/annurev.biochem.72.121801.161800. PMID 12651740.
^ Li M, Brooks CL, Wu-Baer F, Chen D, Baer R, Gu W (Dec 2003). "Mono- versus polyubiquitination: differential control of p53 fate by Mdm2". Science. 302 (5652): 1972–5. Bibcode:2003Sci...302.1972L. doi:10.1126/science.1091362. PMID 14671306.
^ Lipkowitz S, Weissman AM (Sep 2011). "RINGs of good and evil: RING finger ubiquitin ligases at the crossroads of tumour suppression and oncogenesis". Nature Reviews. Cancer. 11 (9): 629–43. doi:10.1038/nrc3120. PMC 3542975. PMID 21863050.
^ ab Hou YC, Deng JY (Jan 2015). "Role of E3 ubiquitin ligases in gastric cancer". World Journal of Gastroenterology. 21 (3): 786–93. doi:10.3748/wjg.v21.i3.786. PMC 4299330. PMID 25624711.
^ de Martino M, Taus C, Wessely IS, Lucca I, Hofbauer SL, Haitel A, Shariat SF, Klatte T (Feb 2015). "The T309G Murine Double Minute 2 Gene Polymorphism Is an Independent Prognostic Factor for Patients with Renal Cell Carcinoma". DNA and Cell Biology. 34 (2): 107–12. doi:10.1089/dna.2014.2653. PMID 25415135.
^ Tang T, Song X, Yang Z, Huang L, Wang W, Tan H (Nov 2014). "Association between murine double minute 2 T309G polymorphism and risk of liver cancer". Tumour Biology. 35 (11): 11353–7. doi:10.1007/s13277-014-2432-9. PMID 25119589.
^ Ardley HC, Robinson PA (2005). "E3 ubiquitin ligases". Essays in Biochemistry. 41: 15–30. doi:10.1042/EB0410015. PMID 16250895.
^ Bai C, Sen P, Hofmann K, Ma L, Goebl M, Harper JW, Elledge SJ (Jul 1996). "SKP1 connects cell cycle regulators to the ubiquitin proteolysis machinery through a novel motif, the F-box". Cell. 86 (2): 263–74. doi:10.1016/S0092-8674(00)80098-7. PMID 8706131.
External links
Quips article describing E3 Ligase function at PDBe
Ubiquitin-Protein+Ligases at the US National Library of Medicine Medical Subject Headings (MeSH)
EC 6.3.2.19